 
Analysis of the genotypes in the samples usually involves sizing of the amplification products by gel electrophoresis. Variable Number of Tandem Repeats (VNTR) PCR targets repetitive areas of the genome that exhibit length variation. Variations in VNTR lengths in six individuals Multiplex PCR has also been used for analysis of microsatellites and SNPs. Multiplex Ligation-dependent Probe Amplification ( MLPA) permits multiple targets to be amplified using only a single pair of primers, avoiding the resolution limitations of multiplex PCR. In the standard protocol for DNA fingerprinting, the targets assayed are often amplified in groups of 3 or 4. For example, in testing for genetic mutations, six or more amplifications might be combined. This permits the simultaneous analysis of multiple targets in a single sample. Multiplex-PCR uses several pairs of primers annealing to different target sequences. Often only a small modification needs to be made to the standard PCR protocol to achieve a desired goal: This article summarizes many of the most common variations currently or formerly used in molecular biology laboratories familiarity with the fundamental premise by which PCR works and corresponding terms and concepts is necessary for understanding these variant techniques. The versatility of polymerase chain reaction (PCR) has led to modifications of the basic protocol being used in a large number of variant techniques designed for various purposes. Touchdown amplification protocol for short ( 100-2,000 bases) amplicons:ġ cycle at 95☌ 90 sec 10 cycles for 95☌ (15 sec) and 72☌ (10-60 sec) 20-25 cycles for 95☌ (15 sec) and 55-65☌ (10 sec) and 72☌ (10-60 sec) final extension step 72☌ 5 min.Various uses and techniques of polymerase chain reaction in the laboratory 
The PCR machine was programmed for amplification short and long ( 2,000-6,000 bases) amplicons:ġ cycle at 95☌ 90 sec 25-32 cycles for 95☌ (15 sec), 68-72☌ (60-400 sec) and final extension step 72☌ 5 min.Īmplification protocol for short ( 100-2,000 bases) amplicons:ġ cycle at 95☌ 90 sec 25-30 cycles for 95☌ (15 sec) and 64-72☌ (60 sec) final extension step 72☌ 5 min. The PCR performed in a 25 µl reaction mixture containing 25 ng DNA, 1x ThermoPol® buffer (with 2 mM MgCl 2 or MgSO 4), 20-300 nM of primer (for multiplex PCR, primer combinations – maximum 1 µM is total concentration), 0.2 mM dNTPs, 1 U Taq DNA polymerase and (optionally) additional 0.01U Pfu DNA Polymerase (for long products amplification).Ī polymerases mix consisting of 100-500 units of Taq DNA polymerase with 1 unit of Pfu DNA Polymerase greatly increased efficiently of amplification for long bands and the accuracy of the PCR. The denaturation of genomic DNA is easy with short step at 98☌, 5-10 seconds. This step includes primer binding the target and polymerase extension at once the recommended time for this step is 1 second for each 100 bases of PCR product. PCR steps - the primers binding (usually from 55-60☌) and the polymerase extension (usually from 50☌ to 72☌), we recommend to join into one step as 68-72☌. The optimal annealing temperature for PCR is calculated directly as the value for the primer with the lowest Tm (T m min), our empirical formulae: 
For primer combinations with very different Tm, the optimal annealing temperature was chosen according to lowest Tm primer (primer with CG content higher then 50% is tolerant to wide annealing Ta, from 55☌ up to 72☌). The range of optimal annealing temperature (Ta) was calculated Tm of primer or optionally plus 6-12☌, and in practice PCR efficiency was tested with gradient annealing temperature using MasterCycler Gradient (Eppendorf). PCR reaction can set up in room temperature and performed without hot-start enzymes. 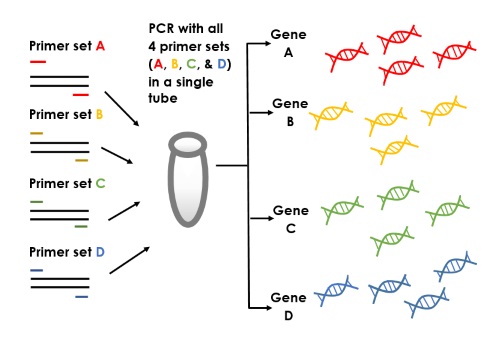
The higher quality of primers is help to save PCR efficiency at changing PCR conditions. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
